Bromine »
PDB 8d90-8gb1 »
8e9e »
Bromine in PDB 8e9e: Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F
Enzymatic activity of Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F
All present enzymatic activity of Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F:
2.5.1.58; 2.5.1.59;
2.5.1.58; 2.5.1.59;
Protein crystallography data
The structure of Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F, PDB code: 8e9e
was solved by
Y.Wang,
Y.Shi,
L.S.Beese,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 43.38 / 2.84 |
| Space group | P 61 |
| Cell size a, b, c (Å), α, β, γ (°) | 169.898, 169.898, 69.339, 90, 90, 120 |
| R / Rfree (%) | 17.6 / 19.4 |
Other elements in 8e9e:
The structure of Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F also contains other interesting chemical elements:
| Zinc | (Zn) | 1 atom |
| Fluorine | (F) | 3 atoms |
Bromine Binding Sites:
The binding sites of Bromine atom in the Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F
(pdb code 8e9e). This binding sites where shown within
5.0 Angstroms radius around Bromine atom.
In total only one binding site of Bromine was determined in the Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F, PDB code: 8e9e:
In total only one binding site of Bromine was determined in the Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F, PDB code: 8e9e:
Bromine binding site 1 out of 1 in 8e9e
Go back to
Bromine binding site 1 out
of 1 in the Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F

Mono view
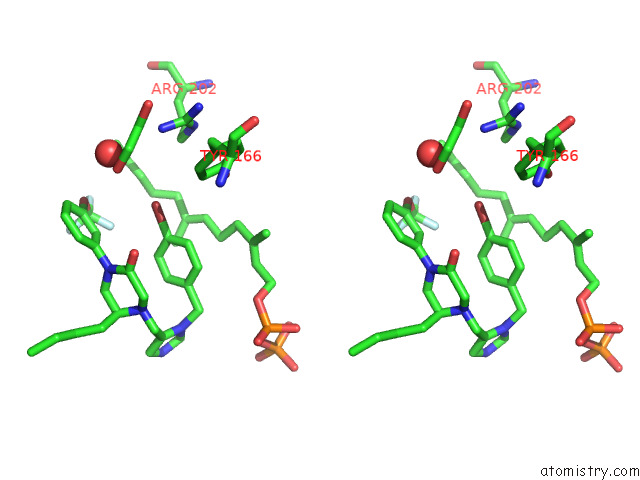
Stereo pair view

Mono view

Stereo pair view
A full contact list of Bromine with other atoms in the Br binding
site number 1 of Rat Protein Farnesyltransferase in Complex with Fpp and Inhibitor 2F within 5.0Å range:
|
Reference:
Y.Wang,
F.Xu,
C.B.Nichols,
Y.Shi,
H.W.Hellinga,
J.A.Alspaugh,
M.D.Distefano,
L.S.Beese.
Structure-Guided Discovery of Potent Antifungals That Prevent Ras Signaling By Inhibiting Protein Farnesyltransferase. J.Med.Chem. V. 65 13753 2022.
ISSN: ISSN 0022-2623
PubMed: 36218371
DOI: 10.1021/ACS.JMEDCHEM.2C00902
Page generated: Mon Jul 7 12:13:14 2025
ISSN: ISSN 0022-2623
PubMed: 36218371
DOI: 10.1021/ACS.JMEDCHEM.2C00902
Last articles
F in 7FAKF in 7F9T
F in 7F9D
F in 7F9Q
F in 7F97
F in 7F9S
F in 7F99
F in 7F9C
F in 7F9A
F in 7F80